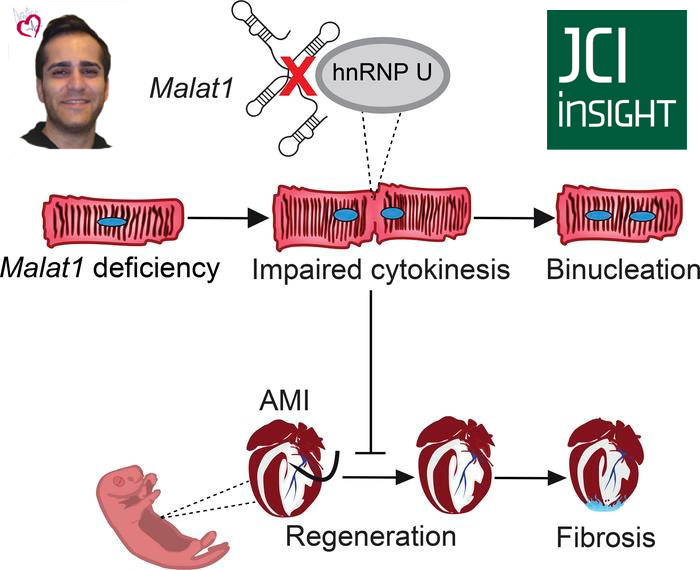
Malat1 deficiency prevents neonatal heart regeneration
The adult mammalian heart has limited regenerative capacity, while the neonatal heart fully regenerates after an insult during the first week of life. In this first week, heart regeneration is mainly driven by proliferation of preexisting cardiomyocytes and supported by pro-regenerative macrophages and angiogenesis. After the first week of life, cardiomyocytes exit the cell cycle and the heart is not able to regenerate anymore. In this work by Aslan and colleagues, we have studied the molecular mechanisms that distinguish regenerative and nonregenerative cardiomyocytes.
We have identified the lncRNA Malat1 as a key player in postnatal cardiac regeneration. Malat1 is highly expressed in neonatal regenerative cardiomyocytes but its expression is reduced in mature nonregenerative cardiomyocytes. Its deletion prevented heart regeneration in mice after myocardial infarction on postnatal day 3 as we could observe larger fibrotic scars in the myocardium. Interestingly, Malat1 deficiency, both in a global and in a cardiomyocyte specific manner, increased cardiomyocyte binucleation even in the absence of cardiac injury. Binucleation is a feature of mature nonregenerative cardiomyocytes.
Furthermore, our work suggests that the interaction of Malat1 and hnRNP U might be required to mediate cardiomyocyte proliferation and binucleation and thus, control the regenerative window in the heart.
https://insight.jci.org/articles/view/162124